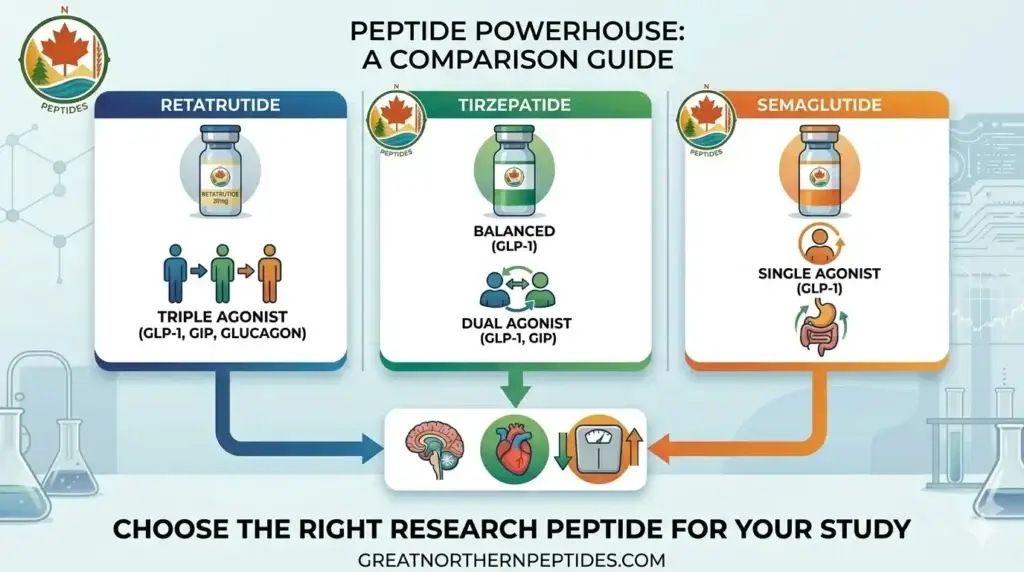
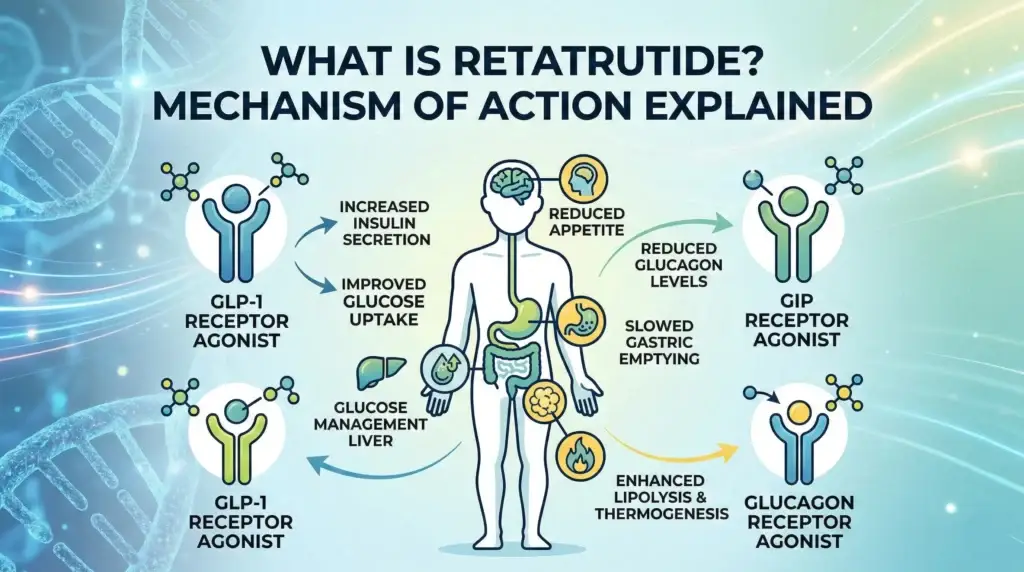
Is Retatrutide Available in Canada? Peptide Research Access Guide
Is Retatrutide Available in Canada? Yes — retatrutide is available in Canada as a research peptide. As it completes Phase 3 clinical trials globally, it is accessible as a research compound for qualified investigators and laboratory use. For a detailed…