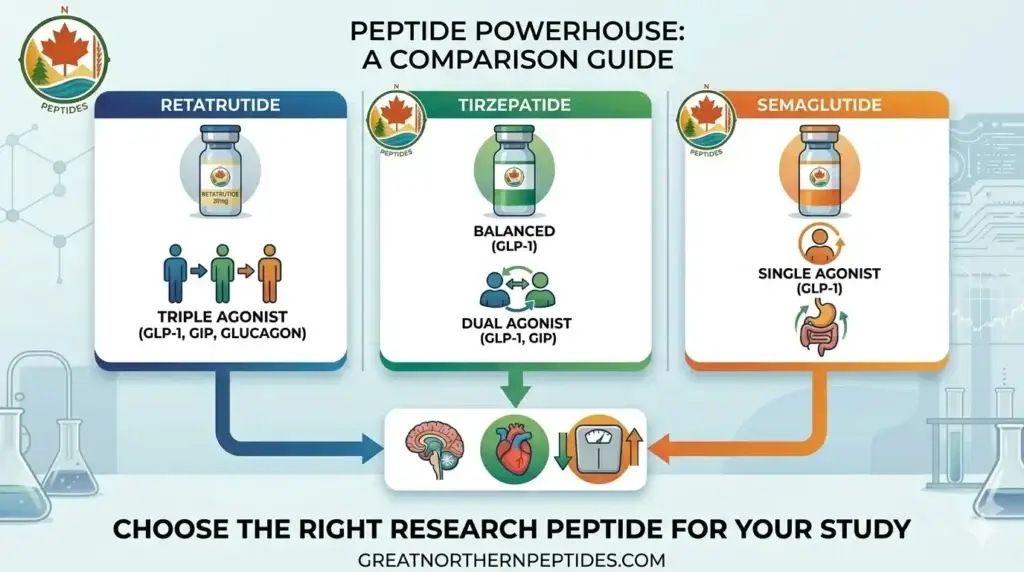
Comparing Retatrutide, Tirzepatide, and Semaglutide
The metabolic peptide research landscape has evolved rapidly with the development of multi-receptor agonists. While semaglutide established the foundation as a single GLP-1 receptor agonist, tirzepatide advanced the field with dual GLP-1/GIP agonism, and now retatrutide represents the next evolution with triple-receptor engagement. This guide breaks down the key differences for researchers.
Receptor Binding Profiles
Semaglutide: Single-Agonist (GLP-1)
Semaglutide selectively targets GLP-1 receptors with high binding affinity. Its albumin-binding fatty acid side chain extends its half-life, enabling once-weekly administration in research protocols. The GLP-1 pathway modulates insulin secretion, gastric motility, and central appetite signalling. Extensive research data, including the STEP trials published in NEJM, have characterized its pharmacological profile thoroughly.
Tirzepatide: Dual-Agonist (GLP-1 + GIP)
Tirzepatide engages both GLP-1 and GIP receptors through a single peptide molecule. The GIP component recruits additional metabolic pathways, particularly in adipose tissue and pancreatic beta cells. The SURMOUNT-1 trial demonstrated that this dual mechanism produced body weight reductions of up to 22.5% — significantly exceeding semaglutide’s single-agonist results.
Retatrutide: Triple-Agonist (GLP-1 + GIP + Glucagon)
Retatrutide adds glucagon receptor agonism to the GLP-1/GIP backbone. The glucagon component activates hepatic lipid oxidation pathways and increases resting energy expenditure, providing a thermogenic dimension absent in both semaglutide and tirzepatide. Learn more in our deep-dive on what retatrutide is and how it works.
Clinical Trial Data Comparison
| Compound | Receptors | Max Weight Reduction (Trial) | Trial Phase | Administration |
|---|---|---|---|---|
| Semaglutide | GLP-1 | ~16.9% (STEP 1, 68 weeks) | Phase 3 / Approved | Once weekly SC |
| Tirzepatide | GLP-1 + GIP | ~22.5% (SURMOUNT-1, 72 weeks) | Phase 3 / Approved | Once weekly SC |
| Retatrutide | GLP-1 + GIP + Glucagon | ~24.2% (Phase 2, 48 weeks) | Phase 2 complete / Phase 3 ongoing | Once weekly SC |
Note: Direct cross-trial comparisons should be interpreted cautiously due to differences in study design, population, and duration. However, the trend toward greater efficacy with broader receptor engagement is consistent across datasets. For a full breakdown of Phase 2 outcomes and the Phase 3 TRIUMPH timeline, see our retatrutide clinical trials 2026 update.
Key Pharmacological Differences
Energy Expenditure
The glucagon receptor component in retatrutide is hypothesized to increase resting energy expenditure through hepatic thermogenesis — a mechanism not present in semaglutide or tirzepatide. Research published in Nature Reviews Endocrinology discusses how glucagon receptor engagement may shift the energy balance equation beyond appetite suppression alone.
Lipid Metabolism
Glucagon receptor activation promotes fatty acid oxidation in the liver, potentially improving hepatic lipid profiles. This is particularly relevant for researchers studying metabolic dysfunction-associated steatotic liver disease (MASLD), where hepatic lipid accumulation is a central pathological feature.
Half-Life and Dosing
All three peptides are engineered for once-weekly subcutaneous administration through structural modifications that extend their circulating half-life. Retatrutide achieves this through a fatty acid moiety similar to the approach used in semaglutide and tirzepatide. For more information look at our dosing guides:
Retatrutide Dosing Guide
Which Peptide for Which Research Application?
- GLP-1 pathway studies → Semaglutide provides a clean, well-characterized single-receptor model
- Dual incretin pathway research → Tirzepatide enables study of GLP-1/GIP synergy
- Multi-receptor metabolic studies → Retatrutide offers the broadest receptor engagement for comprehensive pathway analysis
- Energy expenditure / thermogenesis research → Retatrutide’s glucagon component is uniquely relevant
Canadian researchers can access Retatrutide, Tirzepatide, and Semaglutide for study through Great Northern Peptides, one of the most trusted sources for peptides Canada researchers depend on, with all products third-party tested for purity and identity.
For details on regulatory status and how Canadian researchers access these compounds, see our guide to retatrutide availability in Canada.
Frequently Asked Questions
What is the main difference between retatrutide and tirzepatide?
Tirzepatide is a dual-agonist (GLP-1/GIP), while retatrutide is a triple-agonist that adds glucagon receptor activation. This third receptor target introduces energy expenditure and hepatic lipid oxidation pathways not engaged by tirzepatide.
Is retatrutide more effective than semaglutide?
Phase 2 data showed retatrutide achieving 24.2% body weight reduction vs approximately 16.9% for semaglutide in the STEP 1 trial. However, these were different trials with different designs, so direct comparison requires caution. Phase 3 head-to-head data is not yet available.
Which peptide has the most clinical data?
Semaglutide has the most extensive clinical dataset, including multiple completed Phase 3 trials and regulatory approvals. Tirzepatide also has robust Phase 3 data. Retatrutide has completed Phase 2 with Phase 3 trials ongoing.
Can retatrutide replace tirzepatide in research protocols?
They serve different research purposes. Tirzepatide is preferred for studying dual incretin synergy in isolation, while retatrutide is better suited for studying the additional contribution of glucagon receptor agonism in a multi-receptor context.
Where can I buy retatrutide for research in Canada?
Great Northern Peptides supplies third-party tested retatrutide for Canadian researchers, available in 20mg and 40mg vials with domestic shipping.
{ “@context”: “https://schema.org”, “@type”: “FAQPage”, “mainEntity”: [ { “@type”: “Question”, “name”: “What is the main difference between retatrutide and tirzepatide?”, “acceptedAnswer”: { “@type”: “Answer”, “text”: “Tirzepatide is a dual-agonist (GLP-1/GIP), while retatrutide is a triple-agonist that adds glucagon receptor activation. This third receptor target introduces energy expenditure and hepatic lipid oxidation pathways not engaged by tirzepatide.” } }, { “@type”: “Question”, “name”: “Is retatrutide more effective than semaglutide?”, “acceptedAnswer”: { “@type”: “Answer”, “text”: “Phase 2 data showed retatrutide achieving 24.2% body weight reduction vs approximately 16.9% for semaglutide in the STEP 1 trial. However, these were different trials with different designs, so direct comparison requires caution.” } }, { “@type”: “Question”, “name”: “Which peptide has the most clinical data?”, “acceptedAnswer”: { “@type”: “Answer”, “text”: “Semaglutide has the most extensive clinical dataset, including multiple completed Phase 3 trials and regulatory approvals. Tirzepatide also has robust Phase 3 data. Retatrutide has completed Phase 2 with Phase 3 trials ongoing.” } }, { “@type”: “Question”, “name”: “Can retatrutide replace tirzepatide in research protocols?”, “acceptedAnswer”: { “@type”: “Answer”, “text”: “They serve different research purposes. Tirzepatide is preferred for studying dual incretin synergy in isolation, while retatrutide is better suited for studying the additional contribution of glucagon receptor agonism in a multi-receptor context.” } }, { “@type”: “Question”, “name”: “Where can I buy retatrutide for research in Canada?”, “acceptedAnswer”: { “@type”: “Answer”, “text”: “Great Northern Peptides supplies third-party tested retatrutide for Canadian researchers, available in 20mg and 40mg vials with domestic shipping.” } } ] }