Retatrutide Dosing: A Research Protocol Reference
This guide compiles dosing information, reconstitution procedures, and storage guidelines for retatrutide based on published clinical trial literature and standard peptide handling protocols. It is intended as a reference for researchers working with retatrutide in controlled laboratory settings.
For background on how retatrutide works, see our mechanism of action guide. For broader context on peptide categories, see our types of peptides.
Dosing Schedules from Published Research
Phase 2 Trial Dose Groups
The Phase 2 clinical trial published in NEJM employed the following dose groups, all administered via subcutaneous injection once weekly:
Scroll sideways to view the full table on mobile.
| Dose Group | Escalation Schedule | Maintenance Dose | Duration |
|---|---|---|---|
| Low dose | 0.5mg starting → gradual escalation | 1mg weekly | 48 weeks |
| Medium dose | 0.5mg starting → gradual escalation | 4mg weekly | 48 weeks |
| Medium-high dose | 0.5mg starting → gradual escalation | 8mg weekly | 48 weeks |
| High dose | 0.5mg starting → gradual escalation | 12mg weekly | 48 weeks |
Key observation: All dose groups began at 0.5mg with gradual escalation over several weeks before reaching maintenance doses. This escalation approach is standard across GLP-1 class compounds and serves to improve gastrointestinal tolerability. For a comprehensive summary of these trial results and Phase 3 updates, see our retatrutide clinical trials 2026 update.
Retatrutide Reconstitution Protocol
Lyophilized retatrutide requires reconstitution before use. The following protocol reflects standard peptide reconstitution best practices as outlined by the Health Canada Drug and Health Products and general peptide handling literature:
Required Materials
- Lyophilized retatrutide vial
- Bacteriostatic water (BAC water)
- Insulin syringes (1mL or 0.5mL)
- Alcohol swabs for vial septum sanitation
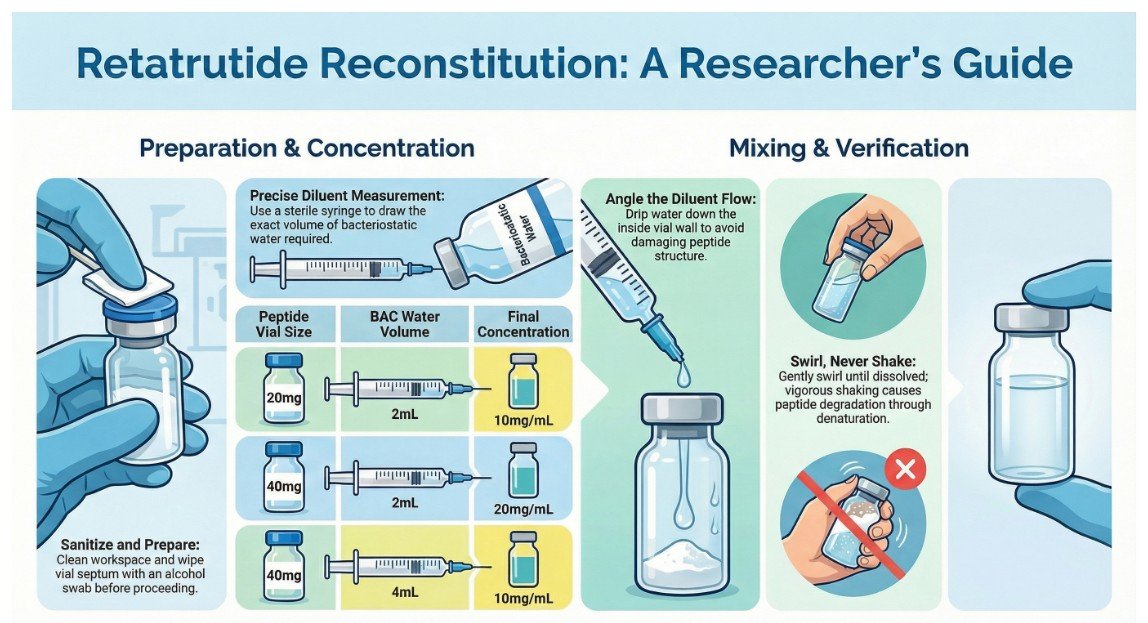
Step-by-Step Reconstitution
- Prepare the workspace: Work in a clean environment. Sanitize hands and work surfaces.
- Clean the vial septum: Wipe the rubber stopper of the peptide vial with an alcohol swab. Allow to air dry.
- Draw diluent: Using a sterile syringe, draw the desired volume of bacteriostatic water. Common reconstitution volumes:
- 20mg vial + 2mL BAC water = 10mg/mL concentration
- 40mg vial + 2mL BAC water = 20mg/mL concentration
- 40mg vial + 3mL BAC water = 13mg/mL concentration
- Add diluent slowly: Insert the needle into the vial at an angle and allow the water to flow down the inside wall of the vial. Do not inject directly onto the lyophilized powder, as this can damage the peptide structure.
- Swirl gently: Once diluent is added, swirl the vial gently until the powder is fully dissolved. Do not shake vigorously — agitation can cause peptide degradation through denaturation.
- Verify dissolution: The solution should be clear and colourless. If particulate matter or cloudiness persists, do not use.
Retatrutide Storage Guidelines
Lyophilized (Pre-Reconstitution)
- Temperature: Store at -20°C for long-term storage; 2-8°C (refrigerator) for short-term
- Light: Protect from direct light
- Stability: Lyophilized peptides are generally stable for 12-24 months when stored appropriately
Reconstituted
- Temperature: Store at 2-8°C (refrigerator) — do not freeze reconstituted peptide
- Stability: Use within 28-30 days when reconstituted with bacteriostatic water
- Note: If reconstituted with sterile water (without benzyl alcohol preservative), use within 24-48 hours and store under strict aseptic conditions
These storage recommendations align with guidelines published by the National Research Council Canada and general pharmaceutical stability literature.
Retatrutide Dosing Calculator




Researchers commonly need to calculate injection volumes based on reconstitution concentration:
Example: 20mg Vial Reconstituted in 2mL BAC Water
- Concentration: 10mg/mL (10,000mcg/mL)
- For a 1mg dose: draw 0.1mL (10 units on a 1mL insulin syringe)
- For a 4mg dose: draw 0.4mL (40 units)
- For a 8mg dose: draw 0.8mL (80 units)
Example: 40mg Vial Reconstituted in 2mL BAC Water
- Concentration: 20mg/mL (20,000mcg/mL)
- For a 4mg dose: draw 0.2mL (20 units)
- For a 8mg dose: draw 0.4mL (40 units)
- For a 12mg dose: draw 0.6mL (60 units)
Research Supplies Available in Canada
Great Northern Peptides provides research-grade retatrutide for Canadian researchers:
- Retatrutide 20mg vials — ideal for lower-dose research protocols
- Retatrutide 40mg vials — suited for higher-dose protocols or longer study durations
- All retatrutide products — browse the full selection
All products include third-party certificates of analysis (COA) verifying purity and identity.
For information on regulatory access in Canada, see our guide to retatrutide availability in Canada.
Frequently Asked Questions
How do you reconstitute retatrutide?
Add bacteriostatic water slowly down the inner wall of the vial containing lyophilized retatrutide. Swirl gently until fully dissolved. Do not shake or inject water directly onto the powder. Common reconstitution volumes are 2mL for a 20mg vial (10mg/mL) or 2-3mL for a 40mg vial.
What is the recommended retatrutide dosage?
Published Phase 2 clinical trial data used doses of 1mg, 4mg, 8mg, and 12mg administered weekly via subcutaneous injection. All groups began at 0.5mg with gradual escalation. Specific dosing should follow institutional research protocols.
How should retatrutide be stored?
Store lyophilized retatrutide at -20°C (long-term) or 2-8°C (short-term). Once reconstituted with bacteriostatic water, store at 2-8°C and use within 28-30 days. Do not freeze reconstituted peptide.
How long does reconstituted retatrutide last?
When reconstituted with bacteriostatic water (which contains benzyl alcohol as a preservative), retatrutide solution is generally stable for 28-30 days when stored at 2-8°C. If sterile water without preservative is used, use within 24-48 hours.
Where can I buy retatrutide pens in Canada?
Retatrutide is currently supplied as lyophilized powder in vials for reconstitution. Canadian researchers can access retatrutide vials from Great Northern Peptides — trusted peptides Canada labs rely on — available in 20mg and 40mg formats with domestic shipping.

 0.3 ml
0.3 ml
 0.5 ml
0.5 ml
 1.0 ml
1.0 ml